The solvent evaporator for fast and easy evaporation of DMSO, DMF, and water.
BioChromato’s Smart Evaporator™ saves time and cost by removing solvents quickly and easily. DMSO, DMF and even water are easily removed without the risk of bumping.



*1 Total worldwide installations of all Smart Evaporator™ models in universities, public institutions, and private companies. Two hundred installations in the USA. (as of April 2023)
*2 The 42nd Invention Award in 2017 (the Japan Society for the Advancement of Inventions)
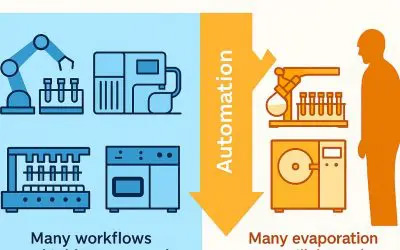
Why “Smart Evaporator™”?
DMSO / DMF / water Evaporator

The Smart Evaporator enables efficient evaporation of high boiling point solvents. With the inert gas purging feature (ex. nitrogen), it’s great for drying down solvents such as DMSO, DMF, toluene, PGMEA and water.
(inert gas recommended).
Learn More
> Application Example of drying water
Bump Free Technology
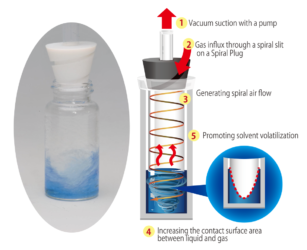
The novel Spiral Plug technology generates a helical flow of air or inert gas over the surface of your solvent, thereby increasing the surface area. Since the vial isn’t under high vacuum, there is no risk of bumping or splashing.
In Vial Evaporation
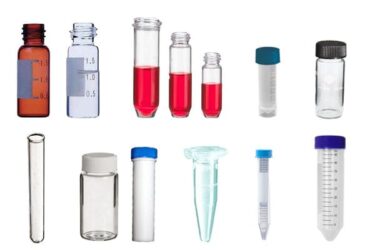
The Smart Evaporator enables The Spiral Plugs come in various sizes for compatibility with many different sample tubes, flasks, and vials. Coupled with a quick release, the Spiral Plugs are easily exchangeable between different sizes.
Explore Smart Evaporator’s Cutting-edge Technology
Learn how Smart Evaporator™ works
Models
Testimonials from our valuable customers
Thank you for all your valuable testimonials!
You can read how they like using the Smart Evaporator.


“A great bit of kit that helped this project proceed at a tremendous pace”
Dr Andrew McGown, Research Fellow in Medicinal Chemistry, Sussex Drug Discovery Centre (SDDC), University of Sussex
>>Read more
“Faster evaporations of extractions and less monitoring of the process!”
Mr. Kasel Nicholas from Donaldson Filtration Solutions
>>Read more
“I am eternally grateful for this device and all of the time it has saved me”
Ms. Teodora “Tia” Nedic, PhD Student Pharmaceutical Sciences, University of California Irvine
>>Read more
Transform Your Laboratory Experience with BioChromato’s Smart Evaporator
Discover the Future of Evaporation
Experience the Difference
Publications
Poster/Application
- Smart Evaporator Verification of Enzyme Activity
- Rapid concentration of high boiling point solvents using a vacuum-assisted vortex concentration method
- Container characteristics during drying and concentration
Introduction Video
Click here to explore more videos about the Smart Evaporator™.
Transform Your Laboratory Experience with BioChromato’s Smart Evaporator