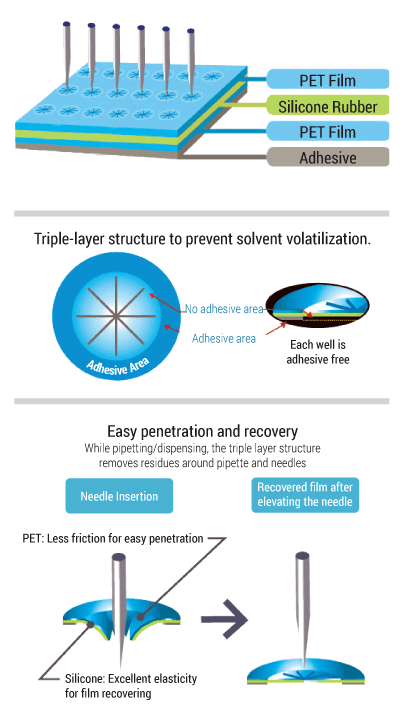
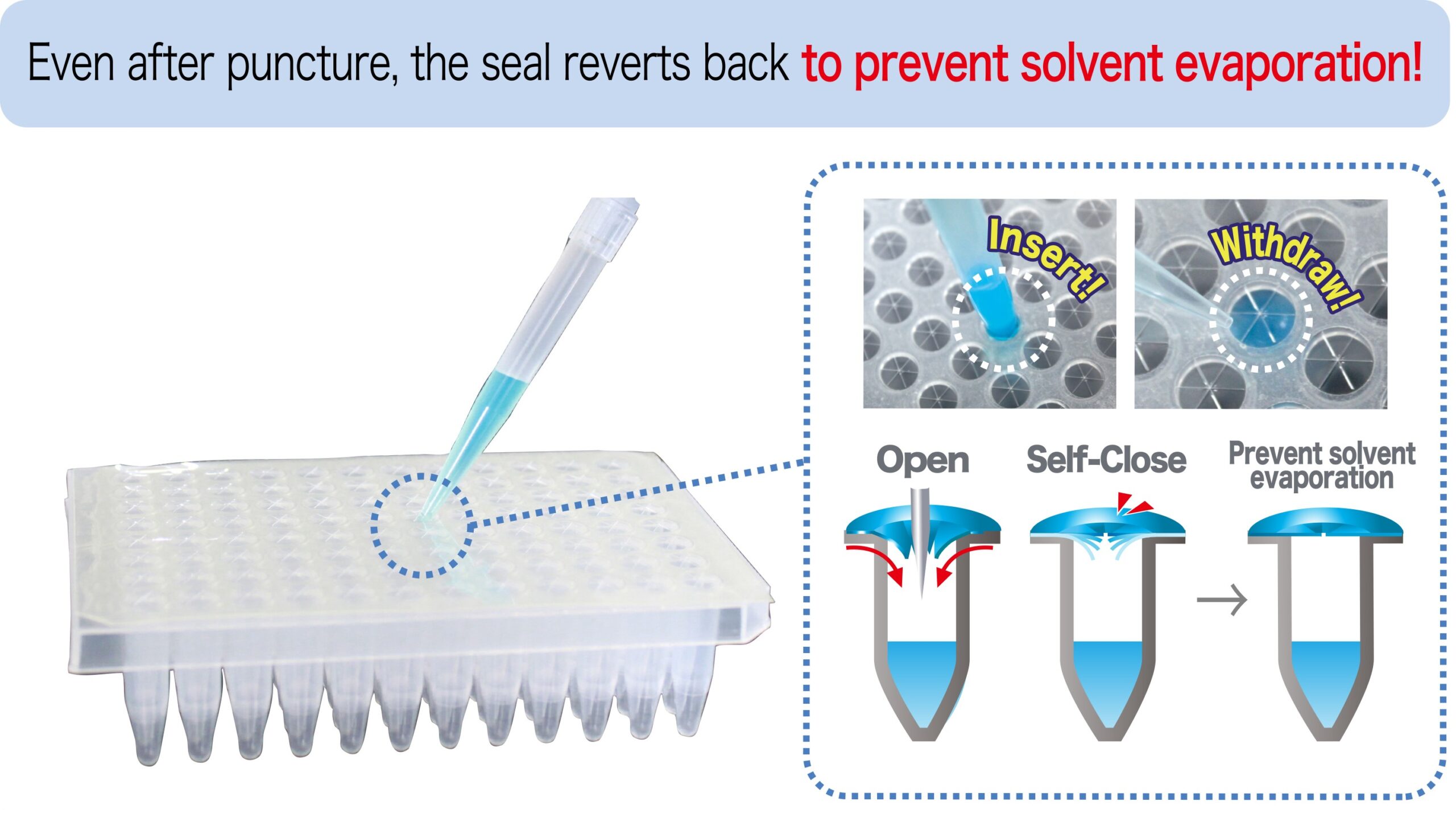
RAPID Slit Seal: A self-sealing plate solution for sample protection.
The RAPID Slit Seal is proven to prevent solvent evaporation and cross-contamination. It allows easy insertion and withdrawal of pipette tips and sampling needles without catching and dragging.
- Self-closes instantly.
- No cross-contamination between wells.
- Allows next day analysis.
- No adhesive on well spots.
- Pipette tips insert easily with less friction.
- Reduce solvent evaporation.
Product Introduction Video
“We run quite a lot of samples with our mass spec, which is why some people in our lab prefer to use the autosampler plate for the samples so that they can just run 96 samples in one sitting…The plate seals are working perfectly for our application.”
Testimonial from Danica-Mae Manalo, Proteomics Lab, Cedars-Sinai Medical Center
Product Summary
Features
- Self-closes instantly
- No cross-contamination between wells
- Allows next day analysis
- No adhesive on well spots
- Pipette tips insert easily with less friction
- Reduce solvent evaporation
Applications
- Automated Single Molecule Imaging
- Cell Culturing
- BioChemical Assays
- Incubation
- Vortex
- Automated Pipetting and Liquid
- Handling
- SPE
- HPLC and LC/MS
- ADME
- DNA Extraction
Specifications
- Size: 80 mm x 122 mm (96-well format)
- Material: PET, Silicone
- Adhesive: Acrylic based (no adhesive over well spots)
- Functional temperature range: -80°C to +100°C
- No PCR inhibition
- If DNA-free certification is needed, please contact us
Reach out now, and we’ll guide you to the perfect application tailored to your needs.

Differences from Silicone Mats:
- Pre-cut slits open easily, and PET layer reduces friction for very easy handling.
- Lower cost than silicone mats.
Very Effective at Preventing Evaporation:
- After 22 hours, 95% of 150 µL water will remain at 37°C
- After 22 hours, 95% of 150 µL of Acetonitrile will remain at 4°C